Omega Plastics Group are an injection moulding full service provider to the medical sector, manufacturing and supplying moulded components and sub-assemblies, as well as direct supply of tooling to customers with their own moulding capability. Offering a full turnkey service taking projects from concept through to full production, with moulding and tool manufacture in one facility.

What does OPG do for the Medical sector?
With our large UK toolroom located on the same site as our moulding facilities it allows full tool and process development and validation (IQ, OQ, PQ) in the same building.
Our involvement in projects starts at design for manufacture stage and we work in partnership with our customers to develop the most appropriate tooling and moulding supply solution. Supporting product evolution and delivering tooling and components for alpha and beta prototype phases, through to supplying higher volume production intent tooling and components for clinical trials, and then commitment to production tooling and scheduled supply of production components.
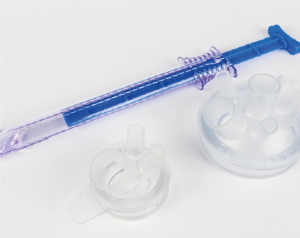
Projects supported:
Working with a diverse range of customers in the Medical sector, supplying components and tooling for the manufacture of: drug delivery devices, surgical devices, implants, bio-reactors, single use/disposables for vaccine manufacture, respiratory products, prosthetics and PPE.

Case Study – Advanced Surgical Concepts
The development of the ASC TriPort & QuadPort is a multi-instrument access port for laparoscopicsurgery used throughout USA & Europe. Advanced Surgical Concepts (ASC) approached Omega Plastics Group with a medical device product that other moulding and design companies were unable to tool.

The products, that are used for Laparoendoscopic Single Site Surgery (LESS), was intended to be manufactured and validated at Omega before transfer to the USA for high volume manufacture. However, due to demand for the device and the successful manufacture within Omega’s ISO Class 8 Cleanroom Omega supported product launch in the USA.
Working together, Omega and the product designer from ASC manufactured a complex single cavity hand loaded mould tool which allowed Omega to manufacture the final product, which ASC used to develop, test and validate the device.
During this time Omega developed a number of variants of the device and moved to multi-cavity tools to further support ramp up and supply. Due to the success of tool development, a product which was initially quoted for initial samples for testing, was successfully used for launch of a fully validated surgical device within the USA and Europe.
This case study highlights how Omega Delivered success where other companies stated, “things could not be done”, a great result for both parties.